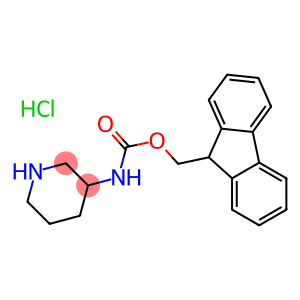
3-N-FMOC-AMINO-PIPERIDINE HCL
9H-fluoren-9-ylmethyl piperidin-3-ylcarbamate hydrochloride
CAS: 672310-13-3
Molecular Formula: C20H23ClN2O2
3-N-FMOC-AMINO-PIPERIDINE HCL - Names and Identifiers
| Name | 9H-fluoren-9-ylmethyl piperidin-3-ylcarbamate hydrochloride |
| Synonyms | 3-N-FMOC-AMINO-PIPERIDINE HCL 3-Fmoc-aminopiperidine hydrochloride 3-N-Fmoc-amino-piperidine hydrochloride (9H-Fluoren-9-yl)methylpiperidin-3-ylcarbamatehydrochloride 9H-fluoren-9-ylmethyl piperidin-3-ylcarbamate hydrochloride 3-Piperidinylcarbamic acid 9H-fluoren-9-ylmethyl ester monohydrochloride Carbamic acid, 3-piperidinyl-, 9H-fluoren-9-ylmethyl ester hydrochloride Carbamic acid, 3-piperidinyl-, 9H-fluoren-9-ylmethyl ester, monohydrochloride (9CI) |
| CAS | 672310-13-3 |
| InChI | InChI=1/C20H22N2O2.ClH/c23-20(22-14-6-5-11-21-12-14)24-13-19-17-9-3-1-7-15(17)16-8-2-4-10-18(16)19;/h1-4,7-10,14,19,21H,5-6,11-13H2,(H,22,23);1H |
3-N-FMOC-AMINO-PIPERIDINE HCL - Physico-chemical Properties
| Molecular Formula | C20H23ClN2O2 |
| Molar Mass | 358.86 |
| Boling Point | 520.4°C at 760 mmHg |
| Flash Point | 268.5°C |
| Vapor Presure | 6.28E-11mmHg at 25°C |
3-N-FMOC-AMINO-PIPERIDINE HCL - Risk and Safety
| HS Code | 29339900 |
3-N-FMOC-AMINO-PIPERIDINE HCL - Introduction
9H-fluoren-9-ylmethyl piperidin-3-ylcarbamate hydrochloride, also known as 9H-fluoren-9-ylmethyl piperidin-3-ylcarbamate hydrochloride, is an organic compound. Its properties are as follows:
-Appearance: Colorless or light yellow solid
-Melting point: about 180-185°C
-Solubility: Soluble in common organic solvents, such as dimethyl sulfoxide, dichloromethane
9H-fluoren-9-ylmethyl piperidin-3-ylcarbamate hydrochloride commonly used in organic synthesis, with the following purposes:
-As a protecting group in organic synthesis: Its Fmoc protecting group can prevent non-specific functional groups in other reactions from reacting through amino protection, thereby playing a protective role in synthesis.
-Used to synthesize peptides: 9H-fluoren-9-ylmethyl piperidin-3-ylcarbamate hydrochloride can be used to synthesize amino acids in peptides or proteins.
The method for synthesizing 9H-fluoren-9-ylmethyl piperidin-3-ylcarbamate hydrochloride is as follows:
1. First, mix 9H-fluorene-9-methylpiperidine and dimethyl sulfoxide (DMSO), and use a sulfonyl cyclization reagent to convert it into an ester.
2. ammonia is added to the cyclized product to hydrolyze the ester bond with 9H-fluorene -9-methyl piperidine to obtain 3-amino -9H-fluorene -9-methyl piperidine.
3. Finally, 3-amino-9H-fluoren-9-methylpiperidine was reacted with FmocCl to give 9H-fluoren-9-ylmethyl piperidin-3-ylcarbamate hydrochloride.
Regarding safety information, the specific toxicity and dangers of 9H-fluoren-9-ylmethyl piperidin-3-ylcarbamate hydrochloride have not been studied in detail. However, when handling any organic compounds, appropriate laboratory safety measures should be taken, such as wearing gloves, goggles and laboratory coats, ensuring that the laboratory is well ventilated, and complying with laboratory safety rules and regulations. When using and handling this compound, the relevant chemical safety practice guidelines should be followed.
-Appearance: Colorless or light yellow solid
-Melting point: about 180-185°C
-Solubility: Soluble in common organic solvents, such as dimethyl sulfoxide, dichloromethane
9H-fluoren-9-ylmethyl piperidin-3-ylcarbamate hydrochloride commonly used in organic synthesis, with the following purposes:
-As a protecting group in organic synthesis: Its Fmoc protecting group can prevent non-specific functional groups in other reactions from reacting through amino protection, thereby playing a protective role in synthesis.
-Used to synthesize peptides: 9H-fluoren-9-ylmethyl piperidin-3-ylcarbamate hydrochloride can be used to synthesize amino acids in peptides or proteins.
The method for synthesizing 9H-fluoren-9-ylmethyl piperidin-3-ylcarbamate hydrochloride is as follows:
1. First, mix 9H-fluorene-9-methylpiperidine and dimethyl sulfoxide (DMSO), and use a sulfonyl cyclization reagent to convert it into an ester.
2. ammonia is added to the cyclized product to hydrolyze the ester bond with 9H-fluorene -9-methyl piperidine to obtain 3-amino -9H-fluorene -9-methyl piperidine.
3. Finally, 3-amino-9H-fluoren-9-methylpiperidine was reacted with FmocCl to give 9H-fluoren-9-ylmethyl piperidin-3-ylcarbamate hydrochloride.
Regarding safety information, the specific toxicity and dangers of 9H-fluoren-9-ylmethyl piperidin-3-ylcarbamate hydrochloride have not been studied in detail. However, when handling any organic compounds, appropriate laboratory safety measures should be taken, such as wearing gloves, goggles and laboratory coats, ensuring that the laboratory is well ventilated, and complying with laboratory safety rules and regulations. When using and handling this compound, the relevant chemical safety practice guidelines should be followed.
Last Update:2024-04-09 21:01:54
Supplier List
Product Name: 3-N-FMOC-AMINO-PIPERIDINE HYDROCHLORIDE Request for quotation
CAS: 672310-13-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 672310-13-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 3-N-FMOC-AMINO-PIPERIDINE HYDROCHLORIDE Visit Supplier Webpage Request for quotation
CAS: 672310-13-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 672310-13-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 3-N-FMOC-AMINO-PIPERIDINE HYDROCHLORIDE Request for quotation
CAS: 672310-13-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 672310-13-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 3-N-FMOC-AMINO-PIPERIDINE HYDROCHLORIDE Visit Supplier Webpage Request for quotation
CAS: 672310-13-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 672310-13-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

